It is easy
to look at the atmosphere and assume that not much is happening but the fact of the matter is the atmosphere is very complex with many layers and contains particulate matter ranging in a variety of sizes and properties. Relatively speaking it is easier to study the atmosphere using bigger particles such as dust or pollen because they can act as a microscopic environment for gaseous contaminants to interact with each other.
Understanding these interactions can help us assess the true geographical effects of atmospheric pollutants from urbanized centres and the extent of natural processes from the surrounding ecosystem in counteracting the imbalance. The two major gaseous contaminants that are widely studied are SO2 (sulphur oxide, we will also refer it as sulphate the ionic derivative) and NOx (Nitrogen Oxide, we will also refer to them as nitrates, the ionic derivative).
In 1990, The Clean Air Act [i] was brought in to reduce the emission of SO2(g) and NOx within the transportation sector. A study by Holland M. D et al. [1999] [ii] on data collected from CASNet (Clean Air Skies Network) concluded a gradual and significant decrease in SO2, and a moderate decrease in NOx after 1990. Our study will extend the study to 2013 with analysis of the characteristics of particle solid phases.
Due to time constraints, we only studied six sites that geographically span across the United States (figure 1). Within western United States Glacier NP and Grand Canyon NP represent less industrialized areas, where as Penn State and Shenandoah NP are within close proximity of industrialized areas. Perkinstown does not have a strong influence from urbanization however, it is located within the Northern Plains Region which has the highest agricultural production as stated by USDA (United States Department of Agriculture) [iii].
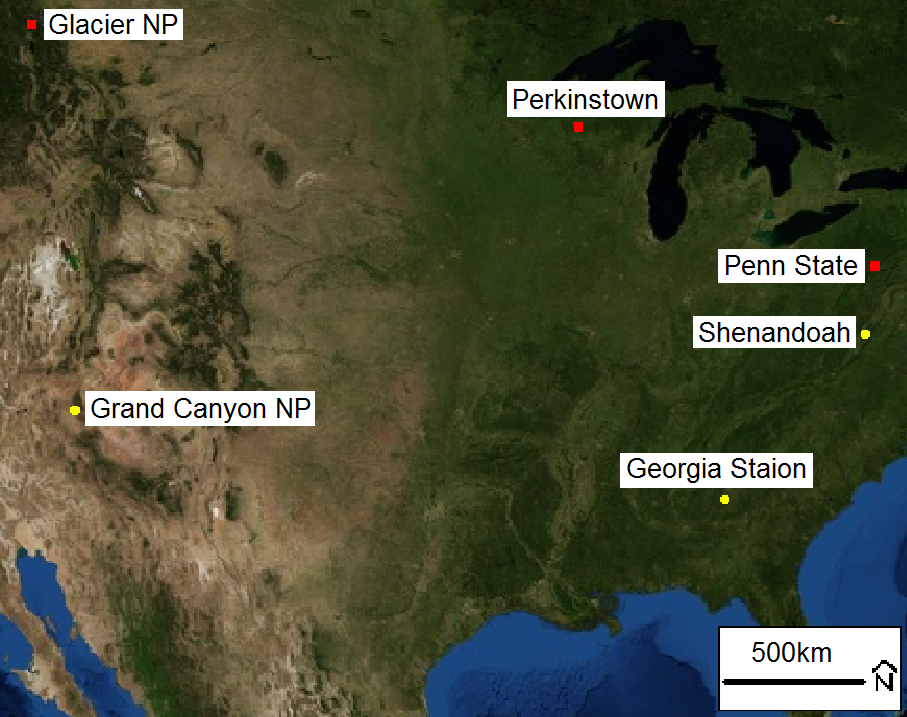
Averages of ion concentration within particulate matter are in alignment with the relative urbanization surrounding the sites, showing that more industrialized areas have higher emissions of sulfates and nitrates. The parameters used to understand overall acidity of the atmosphere are the measurement of strong acidity and for overall neutrality, neutralization. The equations for these parameters can be found in the methodology below. These parameters takes the simplistic approach in estimating the charge balance within the system.
Measurements of the concentrations of sodium Na+ and chloride Cl- were available after 2000, even though their concentrations are relatively small and will not affect the overall annual trends, inclusion in statistical modeling have shown significance. Especially with chloride which has a strong affinity for ammonium creating a strong influence on the neutralization of the system.
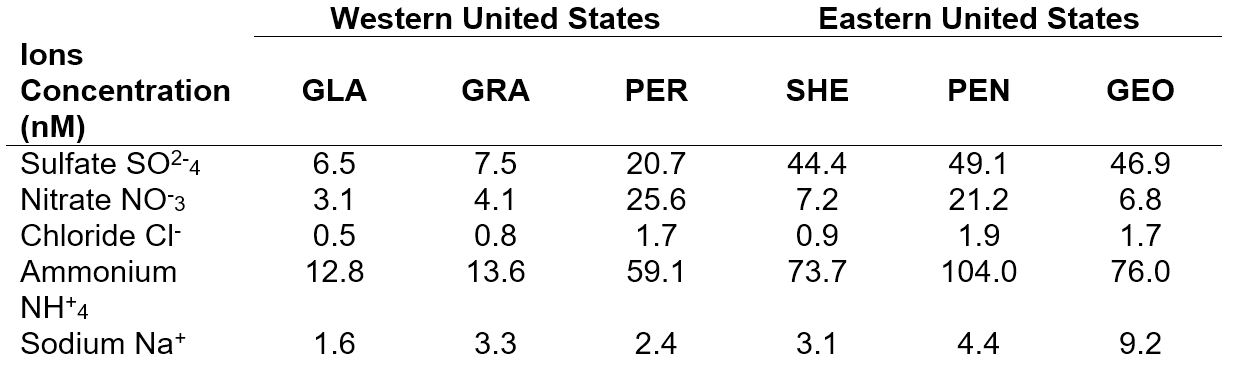
Naturalization of the atmosphere is important because it helps to maintain the pH level of rain water to about 5.6, which is the natural pH of rain. Acidity levels of acid rain below the pH of 5.6 will lead to harmful effects on human health, terrestrial and aquatic ecosystems. Calculations used to determine the acidity of the environment require complex calculations, which were conveniently available as an online model, known as E-AIM (Extended Aerosol Inorganic Model) [iv].
A drawback to our analysis was that only weekly measurements were available. This may not greatly affect the ion concentrations, however weekly averages of relative humanity for E-AIM are completely useless. For the spirt of understanding the particulate system, we analyzed the behaviour of the system by setting the relative humanity at 70%, 80% and 90%. Due to the extensive procedure, only concentrations of Georgia Station were analyzed.
Figure 2 shows the calculated pH values from the E-AIM model. Average pH for rainfall within the United State is about 4.3 by the EPA (Environmental Protection Agency). The wave-like pattern is a result of seasonal changes; during warmer seasons vegetation will naturally produce more ammonia resulting in a lower pH.
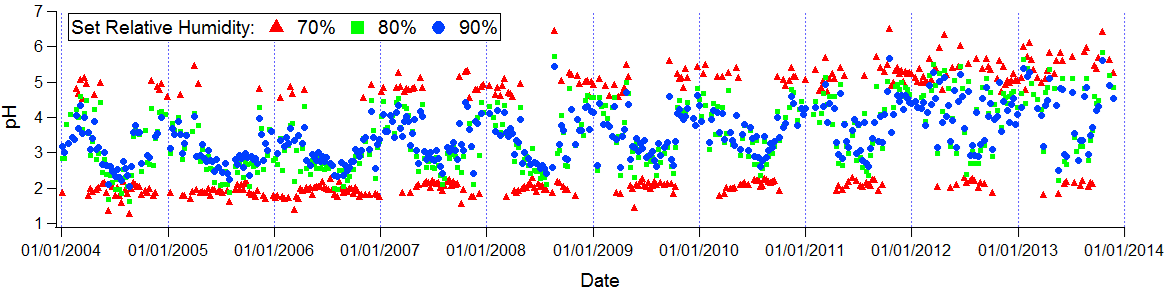
Observations at the relative humidity of 70% are interesting as the system only exists at two levels of pH. Further analyses with set relative humidity at increments of 2% from 70% to 90% shows that at a relative humanity of 78%, the system equalizes into a continuous trend.
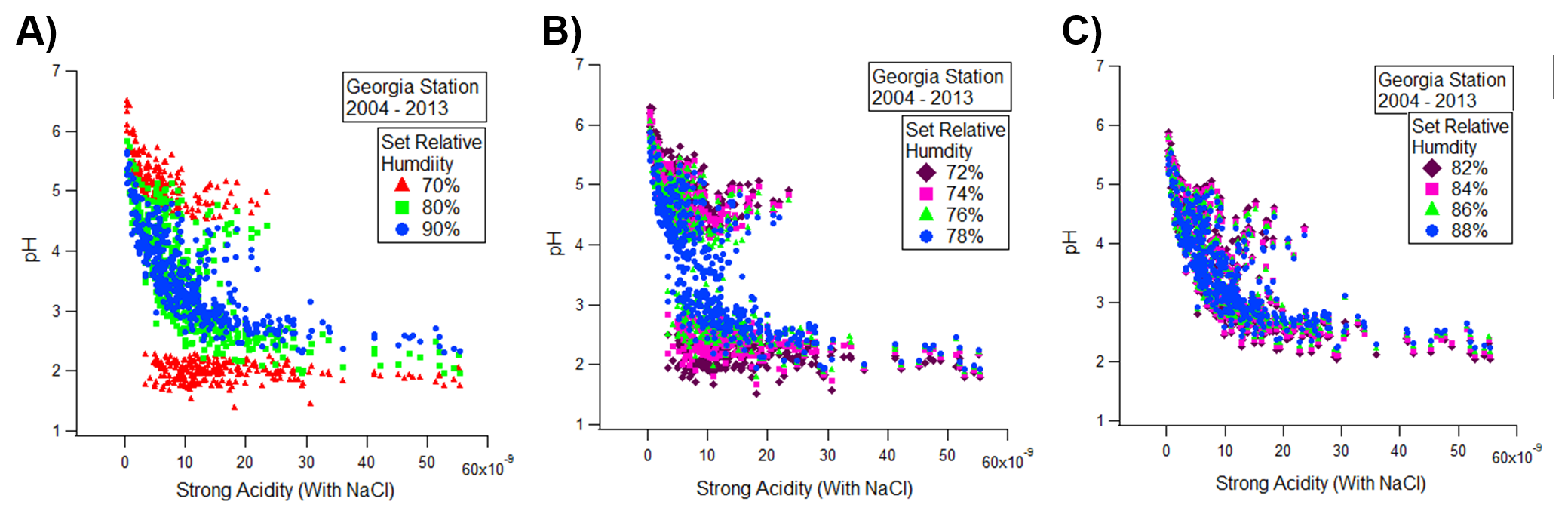
The relative humidity at 78% represents the water activity coefficient, which is the ability for high concentrations of water molecules to enter the particulate phase, in order for the system to have a linear relation between the acidity and ion concentrations.
Our study only covers the interactions with the particulate phase, thus much consideration is placed on understanding the partitioning of water molecules. Relative humidity values are set higher than the average for Georgia Station thus values do not represent the real system. However knowing the strong dependence on relative humidity, we have a better understanding of how to model atmospheric acidity in the future.
Methodology
Measurement and Data
Dry deposition samples were collected weekly by the Clean Air and Skies Network using ion chromatography and coupled plasma-atomic emission spectrometry. The two parameters of interest were strong acidity (equation 1) and the neutralization (equation 2). Concentrations of Na+ and Cl- were available after 2004, thus calculations for the full 20 years (1990-2010) were done without Na+ and Cl-.
Strong acidity:
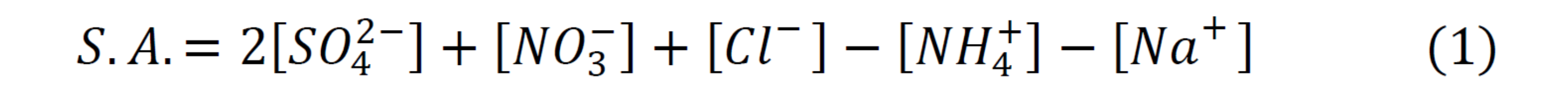
Neutralization:
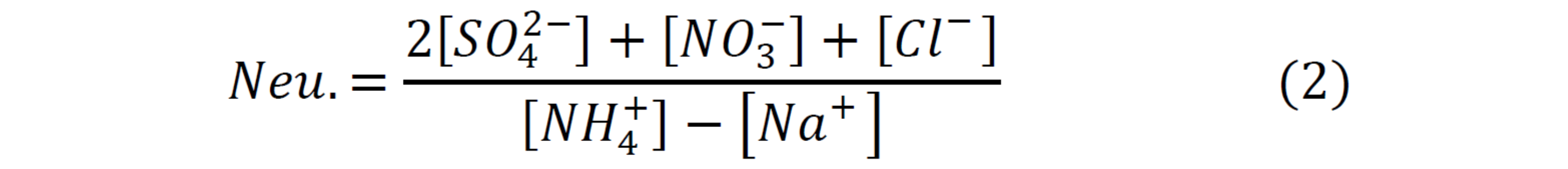
Changes in strong acidity and neutralization were calculated by the Theil-sen slope estimator [v], which calculates the changes of all possible combinations of two ordered-points. An estimation of the slope is calculated by the sum of the scores; +1 when there was in an increase, -1 when there is a decrease and 0 when there is no change. This methodology reduces noise and extreme values.
E-AIM Analysis
Ion concentration from Georgia Station from 2004-2013 was processed using the E-AIM (Extended Aerosol Inorganic Model) [4]. The concentration of H+ (equation 3) was determined to be the ionic equivalent to balance the charges in the system. The model’s outputs were activity coefficients, µ, and molar fractions, n, which were then used to calculate the pH of the system (equation 4).
H+:
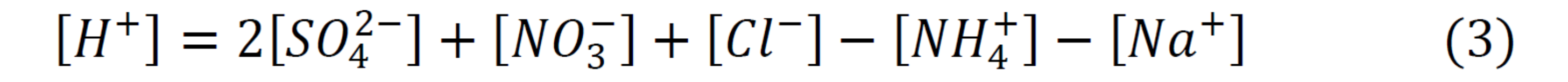
pH:
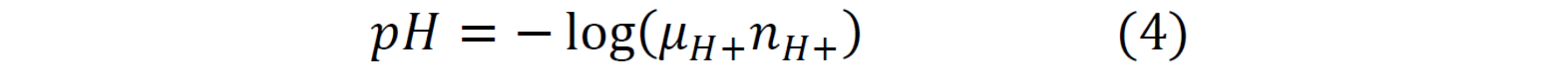
Acknowledgment
Assistance from Professor Murphy and Alex Tevlin is greatly acknowledged and appreciated.
Back to Research
Meet The Team
Reference
i)U.S. Environment Protection Agency (2013) The Clean Air Act Amendments of 1990. Epa.gov/oar/caa/caaa_overview.html, Office of Air and Radiation Communication Office UP EPA. Washington D.C., August 2013.
ii)Holland, D. M., Principe, P. P., Sickles, J. E. II. (1999) Trends in atmospheric sulfur and nitrogen species in the eastern United States for 1989-1995. Atm. Env. 33, 37-49.
iii)Major Uses of Land in the United Sates, 2002. EIB-14. Available from Economic Research Service, USDA United States Department of Agriculture.
iv)E Aim (Extend Aerosol Inorganics Model) (2013) Extended AIM Aerosol Thermodynamic model. www.aim.env.uea.ac.uk/aim/aim.php(accessed 15 March).
v)Theil, H. 1950. A rank-invariant method of linear and polynomial regression analysis. Proc. Kon. Ned. Akad. V. Wetensch. A, 53, 386-392, 521-525, 1397-14-12.